a. Cleavage reaction mechanism
The photoinitiator molecules are excited after absorbing the ultraviolet light energy, and the excited state molecular covalent bonds are broken to generate free radicals, namely:
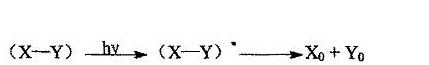
Where (XY) is the ground state initiator molecule, (XY)* is the excited state initiator molecule, and Xo and Yo are the two generated radicals, which may or may not be the same.
b. Mechanism of hydrogen abstraction The mechanism is that the initiator molecules are excited after absorbing light energy, and a hydrogen atom is extracted from monomer or oligomer molecules to make these molecules free radicals. The following formula:
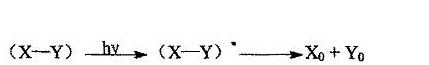
Wherein X is an initiator molecule, X* is an excited state molecule, and P is a monomer or oligomer molecule.
c. Ionic Reaction Mechanism This mechanism is the transfer of electrons and electrons by electron donors and acceptors, which may generate electron transfer complexes and may also generate excited complexes. Electron transfer complexes are formed under ground-state interactions, and the excitation complexes are formed only in the excited state. The mechanism of electron transfer complexes can be expressed as follows:
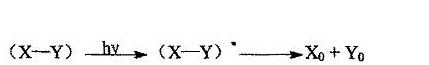
The triplet excited monomer (MTt*) decomposes to generate two radicals, or only a single radical is generated due to electron transfer. The triplet state of thioxanthone has a long life and is a good energy transfer agent. It generates free radicals by the energy transfer mechanism and initiates photopolymerization.

